Paxlovid is dead. Again.
Last month Pfizer’s pill for Covid failed in two large trials published together in the New England Journal of Medicine. The bigger one, with roughly 1,700 participants per group, found 14 people assigned to Paxlovid were hospitalized versus 11 in the control group. No benefit—and leaning in the wrong direction.
This was a long time coming. But it’s definitely been coming.
I’ve written a handful of pieces on Paxlovid, including the hidden studies, nasty side effects, FDA path, and more. Which is why a post-mortem might tell us something about our information ecosystem, and how careful translation of research has the potential to change the world.
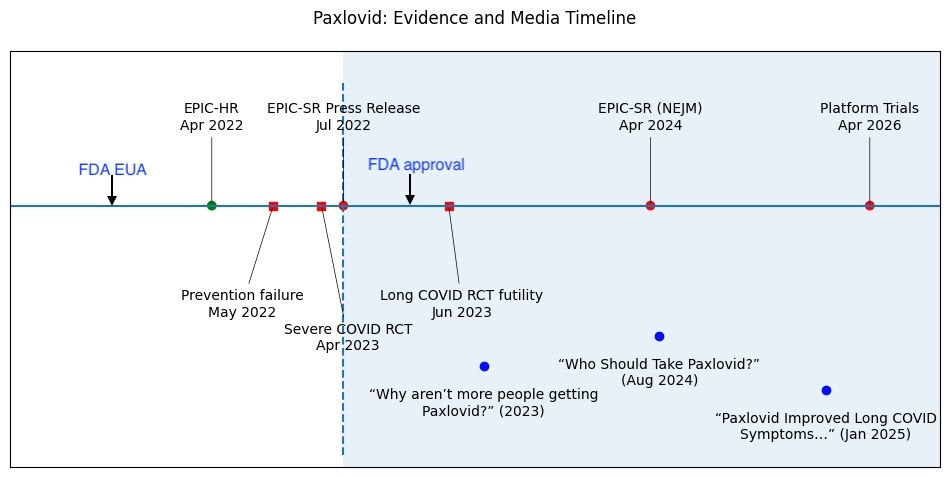
To illustrate, below is a timeline of Paxlovid’s major trials. Green dots indicate a trial that found benefit, red dots mean a trial in which the drug failed.
The first trial, a green dot from 2022, represents the trial that led to the FDA’s Emergency Use Authorization. It was preliminary, rushed, and never intended as a final answer. In it, researchers tested the drug exclusively in people who were unvaccinated and had never been exposed to Covid—a group that no longer exists. In every trial since then, done in populations relevant to today, the drug has failed. The red dots, representing six additional trials, all found no benefit.
To my mind the key moment occurred in July 2022 (the dashed vertical line). This is the date of a press release notifying Pfizer’s investors that Paxlovid’s second trial, which enrolled people vaccinated or previously exposed to Covid, was stopped early for futility. The drug failed to reduce hospitalization or death, and failed to reduce symptoms of Covid. No benefit in any outcome.
This result should have forced everyone to wonder: Can Paxlovid help people in the current world?
The answer was no. Paxlovid failed not just for high-risk people with Covid, it also failed as Covid prevention, for severe Covid, and in Long Covid. By mid-2023 the data were consistent. Worse yet, the evidence for rebound, a relapse illness commonly caused by Paxlovid, was piling up.
With that in mind, now look at the blue dots.
These are NY Times headlines, which were telling a starkly different story. Paxlovid, according to the Times, was beneficial and underused.
Which raises a critical point about medical evidence. For serious research translators, randomized trials are not one input among many. They are at the top of a hierarchy. They are the method we use to strip away bias—to neutralize the hidden distortions that make ineffective treatments appear useful. This is why when randomized trial evidence is available, it replaces weaker forms of evidence. It doesn’t sit beside them. It supersedes them.
And yet with Paxlovid the NY Times repeatedly used weaker studies to rhetorically discredit randomized trials. Their 2025 headline, “Paxlovid Improved Long Covid Symptoms In Some Patients” is shocking, partly because it was based on a 13-person case series. Case series are—literally—the lowest form of scientific evidence available. Two years earlier, Paxlovid had failed in a double-blinded randomized trial for Long Covid.
In fact, just months after the headline, it failed again in a second randomized trial, which the Times neither covered nor even acknowledged.
In this way, quietly, the NY Times inverted the evidence hierarchy, trumpeting weaker studies in order to discredit and eclipse the results of much stronger ones.
But the Times is not alone. They simply co-opted and amplified the opinions of ‘experts’, the CDC, the FDA, the AMA, and more. How did these people and institutions get Paxlovid so wrong? Institutions optimize for their own incentives: Media organizations optimize for engagement. Pharmaceutical companies optimize for sales. Professional societies optimize for relevance and authority. Public health agencies optimize for actionable guidance.
Conventional wisdom is therefore held hostage by the institutions with the power and influence to sculpt it—even when they prefer the opposite of truth. But this is the path to irony, because science will always move toward truth, and falsehoods will be revealed. And when they are the institutions that used their influence for self interest will find that influence slipping away.
Which is how we got here.
The real lesson, therefore, is not that medicine gets things wrong. Of course it does. Urgent first studies of potentially profitable pandemic cures will often be wrong. Think remdesivir, molnupiravir, and others. But rigorous evidence translation sees those errors in real time, and can hold institutions accountable before they dig in—saving them, and the rest of us, from themselves.
Which raises an interesting question for today: The mad scramble to restore institutional influence is now underway—who will blink first? Which major institution will formally correct before the others?
It has been four years since Pfizer announced their drug failed in the only relevant population. Two years since those data were formally published. And two weeks since large new trials beat a dead horse by burying Paxlovid yet again.
As of this writing, perhaps unsurprisingly, the NY Times has offered zero news coverage of the new trials.
Nor is the Times unusual. Yale Medicine still strongly promotes Paxlovid on its public website. Google’s AI overview still says the drug reduces hospitalization and death. The first page of a google search is dominated by FDA, CDC, Pfizer, and Wikipedia entries, all presenting Paxlovid as a lifesaver.
Which helps to clarify the lesson of the Paxlovid timeline.
Bad science is not the greatest danger. Science usually self-corrects. The greater danger is institutional pride and inertia: the years-long gap between when science settles a question and when institutions are willing to absorb the answer.
That gap is costing billions, distorting public understanding, and exposing millions to a harmful drug that simply did not—and does not—work.
But the evidence was there all along.